Instructions For Authors
Northern Clinics of Istanbul (NCI; Istanbul Kuzey Klinikleri) is a double-blind peer-reviewed, open-access, international journal published by the Health Directorate of Istanbul (HDI). NCI is printed six times a year. Free full-text articles in English are available at www.northclinist.com. The journal publishes Research Articles, Case Reports, Letters to the Editor, Review Articles, and Original Images. NCI only accepts manuscripts written in English. Turkish authors should also include an abstract written in Turkish. Opinions presented in published articles do not represent official endorsement of the HDI.
The editorial and publication processes of the journal are shaped in accordance with the guidelines of the International Council of Medical Journal Editors (ICMJE), the World Association of Medical Editors (WAME), the Council of Science Editors (CSE), the Committee on Publication Ethics (COPE), the European Association of Science Editors (EASE), and the National Information Standards Organization (NISO). The journal conforms to the Principles of Transparency and Best Practice in Scholarly Publishing (doaj.org/bestpractice).
Originality, high scientific quality, and citation potential are the most important criteria for a manuscript to be accepted for publication. Manuscripts submitted for evaluation should not have been previously presented or already published in an electronic or printed medium. Manuscripts that have been presented at a conference or other meeting should be submitted with detailed information on the presentation, including the name, date, and location of the event.
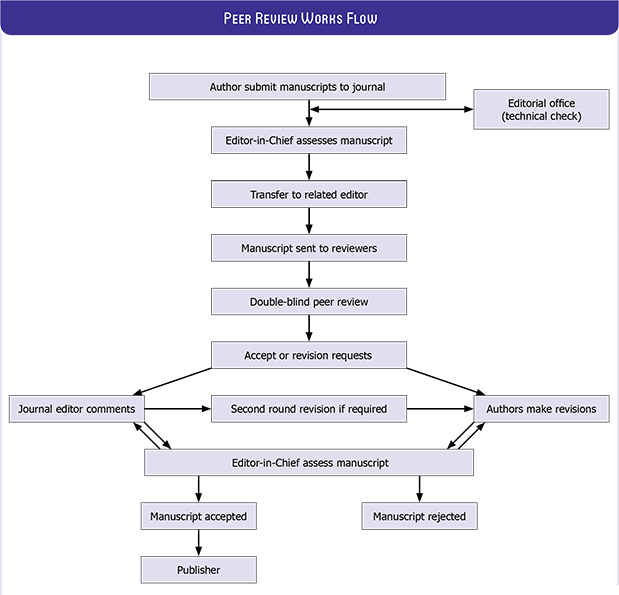
Manuscripts submitted to NCI will go through a double-blind peer-review process. Each submission will be reviewed by at least two external, independent peer reviewers who are experts in their fields in order to ensure an unbiased evaluation process. The editorial board will invite an external and independent editor to manage the evaluation processes of manuscripts submitted by editors or by the editorial board members of the journal. The Editor-in-Chief is the final authority in the decision-making process for all submissions.
Ethics committee approval of research protocols in accordance with international agreements (World Medical Association Declaration of Helsinki “Ethical Principles for Medical Research Involving Human Subjects,” amended in October 2013, www.wma.net) is required for experimental, clinical, and drug studies, and for some case reports. If required, ethics committee reports or an equivalent official document will be requested from the authors. For manuscripts concerning experimental research on humans, a statement should be included indicating that the written, informed consent of patients and volunteers was obtained following a detailed explanation of the procedures that they might undergo. For studies carried out on animals, the measures taken to prevent pain and suffering of the animals should be stated clearly. Information on patient consent, the name of the ethics committee, and the ethics committee approval number should also be stated in the Materials and Methods section of the manuscript. It is the authors’ responsibility to carefully protect the patients’ anonymity. If there is a chance that a patient may be identified from a photograph or other image, or any accompanying text, releases signed by the patient or their legal representative should be enclosed. Identifying images should also be anonymized.
All submissions are screened with similarity detection software (iThenticate by Turnitin).
In the event of alleged or suspected research misconduct, e.g., plagiarism, citation manipulation, or data falsification/fabrication, the Editorial Board will follow up and act in accordance with COPE guidelines.
Article Processing Charge
Starting from 15 April 2024, the Northern Clinics of Istanbul will be introducing an Article Processing Charge (APC) of 400 USD (VAT included) for articles published in the journal. This charge is necessary to cover the costs associated with the publication services provided by the journal. These services include article management costs, the production of articles in PDF and other formats, and the distribution of published articles to various venues and other publishing functions.
This measure is essential to sustain the high-quality standards and operational expenses of the journal. While it represents an additional cost for authors, it is a common practice in scientific publishing, particularly in the context of maintaining the viability of publications like open-access journals.
Statements
All statements and opinions expressed in the manuscripts published in NCI reflect the views of the author(s). All liability for the advertisements rests with the appropriate organization(s). The HDI, the Editor-in-Chief, and Kare Publishing do not accept any responsibility for claims made in articles or advertisements.
The manuscripts submitted to the journal, except abstracts, presentations, reviews, and parts of theses, should not have been accepted and published previously elsewhere in electronic or printed format. Manuscripts evaluated and rejected by other journals must mention the previous submissions and supply reviewer’s reports. This will help to accelerate the evaluation process. If the submitted manuscript has been previously presented at a meeting, the name, date, city and country must be specified.
The authors transfer all copyright to the manuscript to the HDI according to the framework of national and international regulations at the start of the evaluation process. A Copyright Transfer Form signed by the corresponding author must be submitted to the journal with the manuscript. A separate form should be submitted for each manuscript. Manuscripts submitted without a Copyright Transfer Form will not be accepted. After acceptance of the manuscript, all of the authors must complete and sign a Copyright Transfer Form. The authors must affirm that they will not submit the work to another journal, publish it in the original or another language, or allow a third party to use the manuscript without the written permission of the HDI. In the event that the manuscript is rejected, all copyrights transfer back to the authors. Manuscripts that are declined will not be returned, with the exception of artwork.
All content is the authors’ responsibility. All financial liability and legal responsibility associated with the copyright of submitted tables, figures, and other visual materials protected by national and international laws rest with the authors. The authors take responsibility for any legal proceedings issued against the journal.
Declaration of Artificial Intelligence (AI)–Assisted Technology in Scientific Writing
At submission, authors should disclose whether they used artificial intelligence (AI)– assisted technologies (such as Large Language Models [LLMs],
chatbots, or image creators) in the production of submitted work. Authors who use such technology should describe, in both the Title Page and the
submitted work, how they used it. Authors who used AI technology to conduct the study should describe its use in the methods section in sufficient detail to enable replication to the approach, including the tool used, version, and prompts where applicable. Chatbots (such as ChatGPT) and AI assisted technologies should not be listed as an author or co-author nor cited
because they cannot be responsible for the accuracy, integrity, and originality of the work, and these responsibilities are required for authorship.
Therefore, humans are responsible for any submitted material that included the use of AI-assisted technologies.
Authors should carefully review and edit the result because AI can generate authoritative-sounding output that can be incorrect, incomplete, or biased.
Authors should be able to assert that there is no plagiarism in their paper, including in text and images produced by the AI.
Humans must ensure there is appropriate attribution of all quoted material, including full citations.
To clarify scientific contributions and responsibilities and any conflict of interest issues relevant to the manuscript, all parts of the Authors' Contribution Form must be completed by the corresponding author and the ICMJE Uniform Disclosure Form for Potential Conflicts of Interest must be completed online by all authors. Both forms should be included with the manuscript at the time of original submission.
Author names will be published as they are listed on the Copyright Transfer Form. Requests for changes in affiliation or the order of names at a later date cannot be granted to protect all parties involved.
Authors are required to submit the following:
- Copyright Transfer Form,
- Author Contributions Form, and
- ICMJE Potential Conflict of Interest Disclosure Form (should be completed
by all contributing authors) at the time of the initial submission. These forms are available for download at www.northclinist.com
Preparation of the Manuscript
General Format: All manuscripts should be typed using the standard A4-size format document with 2.5 cm-wide margins on all sides.
The references should be numbered consecutively in the order of their first mention in the text.
All text material, including references, footnotes, and table and figure legends, should be typed using double-spacing in an 11 point font with
left alignment and without hyphenated line breaks. The fonts Times New Roman or Arial should be used in the text, for symbols, and all other
special characters. Please use the editing features of your word processing program to type bold or italic letters, mathematical symbols,
Greek letters, subscript and superscript characters. Please take care not to confuse the letters O and I with the numerals 0 and 1.
To set a left indent for a paragraph, click the TAB button once. Only the International System of Units (SI) should be used for units of measurement.
Please review the final version of the manuscript very carefully, especially for formatting and editing errors.
Please note that American English spelling and terminology should be used in the manuscripts.
If the English used in the article is not found to be of sufficient quality, the authors may be required to obtain and provide evidence of
professional assistance.
All pages of the manuscript should be consecutively numbered starting from the first page (page 1, Turkish abstract (Turkish authors only); page 2, English abstract, etc.). Page numbers should be indicated on the upper right-hand corner of each page. The final electronic version of the manuscript should be in “.doc”, “docx” or “.rtf” format. Manuscripts submitted in “.pdf” format will not be accepted.
Accepted manuscripts are copy-edited for grammar, punctuation, format, and clarity. Artificial intelligence technology is partially used in the English language correction of articles. A PDF proof of the manuscript is sent to the corresponding author and their publication approval is requested within 2 days of receipt of the proof.
Title page: A separate title page should be submitted with all manuscripts and this page should include:
- The full title of the manuscript as well as a short title (running head) of no more than 50 characters,
- Name, affiliation(s), ORCID ID, and highest academic degree(s) of the author(s),
- Details of any grant or other sources of support,
- Name, address, telephone (including mobile phone number) and fax numbers, and email address of the corresponding author,
- Acknowledgment of the individuals who contributed to the preparation of the manuscript but who do not fulfill authorship criteria.
- Authors should disclose whether they used artificial intelligence (AI)– assisted technologies (such as Large Language Models [LLMs], chatbots, or image creators) in the production of submitted work. Authors should assert that there is no plagiarism in their paper, including in text and images produced by the AI -if any- and must ensure there is appropriate attribution of all quoted material, including full citations. Authors who used AI technology to conduct the study should describe its use in the methods section in sufficient detail.
Abstract: An abstract should be submitted with all manuscripts with the exception of Letters to the Editor.
The abstract of an Original Article should be structured with subheadings (Objective, Methods, Results, and Conclusion).
All acronyms and abbreviations used in the manuscript should be defined at first use, both in the abstract and in the main text.
The abbreviation should be provided in parentheses following the definition.
Please refer to Table 1 below for word count specifications.
Keywords: Each submission must be accompanied by a minimum of three to a maximum of six keywords at the end of the abstract to be used for subject indexing. The keywords should be listed in full without abbreviations. The keywords should be selected from the National Library of Medicine, Medical Subject Headings database (https://www.nlm.nih.gov/mesh/MBrowser.html).
Introduction: State the specific purpose and available data relevant to the study.
Methods: All methods used to select participants and conduct the study should be described in detail. Known methods should be cited. Novel or modified methods used should be described in detail. Doses, concentrations, routes, and duration of administration of drugs and chemical agents should be indicated. A concise report of all statistical methods used for summarizing available data and for testing the proposed hypothesis should be provided under a subtitle, including the p value criteria determined for statistically significant difference. Statistical evaluation conducted should be explained in detail. Standard statistical methods should be used as much as possible. If rarely employed or novel statistical methods were used, then the relevant references should be cited. When necessary, more detailed explanations about unusual, complex, or new statistical methods can be provided in separate files for readers as online supplementary data. When a trademarked drug, product, hardware, or software program is mentioned within the main text, product information, include the name of the product, the manufacturer of the product, and the city and the country of the company headquarters (including the state if in USA), should be provided in parentheses in the following format: “Discovery St PET/CT scanner (General Electric, Milwaukee, WI, USA). The recommendations in the statistics section of the “Uniform Requirements for Manuscripts Submitted to Biomedical Journals: Writing and Editing for Biomedical Publication” (http://www.ICMJE.org) should be taken into consideration. Authors who used AI technology to conduct the study should describe its use in this section in sufficient detail to enable replication to the approach, including the tool used, version, and prompts where applicable.
Results: The study results should be presented in logical sequence and in detail. The findings should be supported by figures and tables. Information given in figures and tables should not be repeated in the text unless absolutely required.
Discussion: Data relevant to the study subject matter should be examined, evaluated, and substantiated with references from domestic and international sources. General information irrelevant or superfluous to the report should not be included.
Acknowledgement: The names of individuals who contributed to the study but who fail to meet the criteria of authorship should be mentioned in this section. The written consent of all individuals mentioned should be obtained.
Highlight Key Points: Each submission should be accompanied by 3 to 5 “highlight key points” which should emphasize the most striking results of the study and highlight the message that is intended to be conveyed to the readers. These points should be constructed in a way that provides the readers with a general overview of the article and enables them to have a general idea about the article. The highlights should be listed at the end of the main text, above the reference list.
Manuscript Types
NCI publishes the types of articles briefly described below.
Research Articles: This is the most important type of article, since it provides new information based on original research. The main text of original articles should be structured with an Introduction, Methods, Results, Discussion, Conclusion, and References subheadings. Please see Table 1 for limitations for Research Articles.
Statistical analysis is usually necessary to support conclusions. Statistical analyses must be conducted in accordance with international statistical reporting standards (Altman DG, Gore SM, Gardner MJ, Pocock SJ. Statistical guidelines for contributors to medical journals. Br Med J 1983: 7; 1489-93). Information on statistical analyses should be provided with a separate subheading under the Materials and Methods section and the statistical software that was used during the process must be specified.
Units should be prepared in accordance with the International System of Units (SI).
Limitations, drawbacks, and the shortcomings of original articles should be mentioned in the Discussion section before the conclusion paragraph.
Review Articles: Reviews prepared by authors who have extensive knowledge of a particular field and whose scientific background has been translated into a large volume of publications with a high citation potential are welcomed. Submissions from such authors may also be invited by the journal. Reviews should describe, discuss, and evaluate the current level of knowledge of a topic in clinical practice and should guide future studies. The main text should include an Introduction, Clinical and Research Consequences, and Conclusion sections. Please refer to Table 1 for the limitations for Review Articles.
ANNOUNCEMENT FOR AUTHORS
As a large number of case reports are awaiting publication, Northern Clinics of Istanbul has decided to stop acknowledging CASE REPORTS and ORIGINAL IMAGES for a certain period of time until the backlog is cleared. Please DO NOT SUBMIT CASE REPORTS and ORIGINAL IMAGES.
Case Reports: There is limited space for case reports in the journal and reports on rare cases or conditions that constitute challenges in diagnosis and treatment, those offering new therapies or revealing knowledge not included in the literature, and interesting and educative case reports are accepted for publication. The text should include an Introduction, Case Presentation, and Discussion subheadings. Interesting and unusual images are an advantage in the evaluation process. Please see Table 1 for the limitations for Case Reports.
Original Image: Rare and impressive images that reflect significant findings based on clinical science, shed light on fundamental mechanisms of diseases, emphasize abnormalities, or introduce new treatment methods are accepted for publication. The document should be structured with an initial paragraph of a brief case presentation followed by the general information.
Letters to the Editor: This type of manuscript discusses important parts, overlooked aspects, or lacking parts of a previously published article. Articles on subjects within the scope of the journal that might attract the readers’ attention, particularly educative cases, may also be submitted in the form of a Letter to the Editor. Readers can also present their comments on published manuscripts in the form of a Letter to the Editor. An abstract, Keywords, and Tables, Figures, Images, and other media should not be included. The text should not include subheadings. The manuscript that is being commented on must be properly cited within this manuscript.
Table 1. Limitations for each manuscript type
| Type of manuscript | Word limit | Abstract word limit | Reference limit | Table limit | Figure limit |
| Original Article | 3500 | 350 (Structured) | 40 | 6 | 6 |
| Review Article | 5000 | 350 | 50 | 6 | 10 |
| Case Report | 1000 | 200 | 15 | 2 | 5 |
| Case Image | 300 | No abstract | 5 | No tables | 2 |
| Letter to the Editor | 500 | No abstract | 5 | No tables | No media |
Tables
Tables should be included in the main document, presented after the reference list, and they should be numbered consecutively in the order they are referred to within the main text. A descriptive title must be placed above each table. Abbreviations used in the table should be defined below the table by footnotes (even if they are defined within the main text). Tables should be created using the word processing software “insert table” command and they should be arranged clearly to provide easy reading. Data presented in tables should not be a repetition of the data presented within the main text but should support the main text.
Figures and Figure Legends
Figures, graphics, and photographs should be submitted as separate files (in TIFF or JPEG format) through the submission system. The files should not be embedded in a Word document or the main document. When there are figure subunits, the subunits should not be merged to form a single image. Each subunit should be submitted separately through the submission system. Images should not be labeled (a, b, c, etc.) to indicate figure subunits. Thick and thin arrows, arrowheads, stars, asterisks, and similar marks may be used on the images to support the figure legends. Like the rest of the submission, the figures should also be blind. Any information within the images that may identify an individual or institution should be anonymized. The minimum resolution of each submitted figure should be 300 DPI. To prevent delays in the evaluation process, all submitted figures should be clear in resolution and large in size (minimum dimensions: 100 × 100 mm). Figure legends should be listed at the end of the main document.
References
All references, tables, and figures should be referred to within the main text, and they should be numbered consecutively in the order they are referred to within the main text.
While citing publications, preference should be given to the latest, most up-to-date publications. If an ahead-of-print publication is cited, the DOI number should be provided. Authors are responsible for the accuracy of references. Journal titles should be abbreviated in accordance with the journal abbreviations in the Index Medicus/ MEDLINE/PubMed. When there are 6 or fewer authors, all authors should be listed. If there are 7 or more authors, the first 6 authors should be listed followed by “et al.” In the main text of the manuscript, references should be cited using Arabic numbers in parentheses. The reference styles for different types of publications are presented in the following examples.
Journal Article:
Author. Title. Journal Year;Volume:Pages.
Rankovic A, Rancic N, Jovanovic M, Ivanovic M, Gajovic O, Lazic Z, et al. Impact of imaging diagnostics on the budget – Are we spending too much? Vojnosanit Pregl 2013;70:709–11.
Book Section:
Author. Title. In: Editor, `editor`.^`editors`. Book Title. Edition ed. Place Published: Publisher; Year. p. Pages.
Suh KN, Keystone JS. Malaria and babesiosis In: Gorbach SL, Barlett JG, Blacklow NR, editors. Infectious Diseases. 3th ed . Philadelphia: Lippincott Williams; 2004. p.2290–308.
Books with a Single Author:
Author. Title. Edition ed. Place Published: Publisher; Year.
Sweetman SC. Martindale the Complete Drug Reference. 34th ed. London: Pharmaceutical Press; 2005.
Conference Proceedings:
Author. Title. In: Editor, `editor`.^`editors`. Conference Name; Year of Conference Date; Conference Location: Publisher; Year of Conference|. p. Pages.
Bengisson S. Sothemin BG. Enforcement of data protection, privacy and security in medical informatics. In: Lun KC, Degoulet P, Piemme TE, Rienhoff O, editors. MEDINFO 92. Proceedings of the 7th World Congress on Medical Informatics; 1992 Sept 6-10; Geneva, Switzerland. Amsterdam: North-Holland; 1992. pp.1561-5.
Scientific or Technical Report:
Author. Title. Type. Place Published: Institution; Year Date. Report No.: Report Number.
Cusick M, Chew EY, Hoogwerf B, Agrón E, Wu L, Lindley A, et al. Early Treatment Diabetic Retinopathy Study Research Group. Risk factors for renal replacement therapy in the Early Treatment Diabetic Retinopathy Study (ETDRS), Early Treatment Diabetic Retinopathy Study Kidney Int: 2004. Report No: 26.
Thesis:
Author. Title. Type. Place Published: Institution; Year Date. Report No.: Report Number.
Kaplan SI. Post-hospital home health care: elderly access and utilization (dissertation). St Louis (MO): Washington Univ; 1995.
Epub Ahead of Print Articles:
Author. Title. Alternate Title Year Date Accessed. doi: DOI. [Epub ahead of print].
Cai L, Yeh BM, Westphalen AC, Roberts JP, Wang ZJ. Adult living donor liver imaging. Diagn Interv Radiol. 2016 Feb 24. doi: 10.5152/dir.2016.15323. [Epub ahead of print].
Webpage:
Author. Title. Available at: URL. Accessed Access Date, Access Year.
Copyright Notice
All published material will become the sole property and will be copyrighted by the Journal. No part of the publication may be used without prior written permission of the publisher. Articles and illustrations become the property of the Journal after publication.
Open Access and Commons User Licenses
Open Access
The NCI is an open access journal which means that all content is freely available without charge to the user or his/her institution. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author. This is in accordance with the BOAI definition of open access.
Commons User Licenses
This work is licensed under a Creative Commons Attribution-Non Commercial 4.0 International License (CC BY-NC 4.0) .
Ownership and Accountability for Contents on behalf of the Health Directorate of Istanbul
Kemal Memisoglu, M.D.
Editor-in-Chief
Berna Terziođlu Bebitođlu, MD
Department of Medical Pharmacology, Marmara University, Faculty of Medicine, Ýstanbul, Turkey
E-mail: [email protected]
Publisher
KARE PUBLISHINGGöztepe Mah. Fahrettin Kerim Gökay Cad. No: 200 Da: 2, Göztepe, Kadýköy, Ýstanbul
Tel: +90 216 550 61 11
Fax: +90 216 550 61 12
www.karepb.com
E-mail: [email protected]